Scientists find new way to produce hydrogen
Friday, January 23, 2009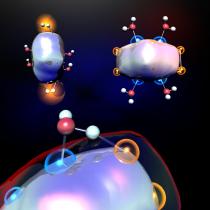 A.C. Reber, VCU/PSU The
figure shows aluminum clusters reacting with water to produce hydrogen.
A.C. Reber, VCU/PSU The
figure shows aluminum clusters reacting with water to produce hydrogen.
University Park, Pa. — Scientists at Penn State and the Virginia Commonwealth University have discovered a way to produce hydrogen by exposing selected clusters of aluminum atoms to water. The findings are important because they demonstrate that it is the geometries of these aluminum clusters, rather than solely their electronic properties, that govern the proximity of the clusters' exposed active sites. The proximity of the clusters' exposed sites plays an important role in affecting the clusters' reactions with water.
The team's findings have been published in the Jan. 23 issue of the
journal Science.
"Our previous research suggested that electronic properties govern
everything about these aluminum clusters, but this new study shows that it
is the arrangement of atoms within the clusters that allows them to split
water," said A. Welford Castleman Jr., Eberly family distinguished chair in
science and Evan Pugh professor in the Penn State Departments of Chemistry
and Physics. "Generally, this knowledge might allow us to design new
nanoscale catalysts by changing the arrangements of atoms in a cluster. The
results could open up a new area of research, not only related to splitting
water, but also to breaking the bonds of other molecules, as well."
The team, which also includes Penn State graduate students Patrick Roach and
Hunter Woodward, and Virginia Commonwealth University Professor of Physics
Shiv Khanna and postdoctoral associate Arthur Reber, investigated the
reactions of water with individual aluminum clusters by combining them under
controlled conditions in a custom-designed flow-reactor. They found that a
water molecule will bind between two aluminum sites in a cluster as long as
one of the sites behaves like a Lewis acid, a positively charged center that
wants to accept an electron, and the other behaves like a Lewis base, a
negatively charged center that wants to give away an electron. The
Lewis-acid aluminum binds to the oxygen in the water and the Lewis-base
aluminum dissociates a hydrogen atom. If this process happens a second time
with another set of two aluminum sites and a water molecule, then two
hydrogen atoms are available, which then can join to become hydrogen gas
(H2).
The team found that the aluminum clusters react differently when exposed to
water, depending on the sizes of the clusters and their unique geometric
structures. Three of the aluminum clusters produced hydrogen from water at
room temperature. "The ability to produce hydrogen at room temperature is
significant because it means that we did not use any heat or energy to
trigger the reaction," said Khanna. "Traditional techniques for splitting
water to produce hydrogen generally require a lot of energy at the time the
hydrogen is generated. But our method allows us to produce hydrogen without
supplying heat, connecting to a battery, or adding electricity. Once the
aluminum clusters are synthesized, they can generate hydrogen on demand
without the need to store it."
Khanna hopes that the team's findings will pave the way toward investigating
how the aluminum clusters can be recycled for continual usage and how the
conditions for the release of hydrogen can be controlled. "It looks as
though we might be able to come up with ways to remove the hydroxyl group
that remains attached to the aluminum clusters after they generate hydrogen
so that we can reuse the aluminum clusters again and again," he said.
The team plans to continue the research with a goal of refining their new
method. This research was supported by the Air Force Office of Scientific
Research.