Researchers have long been trying to discover the mysteries of the brain, as well as target areas within to control behaviors.
For almost 15 years, scientists have been experimenting with
optogenetics, a new way of studying the brain that uses
light to control precise neural activity in freely moving
animals. The challenge has been delivering light to exact
regions deep in the brain. Many tests involved invasive
procedures, embedding fiber-optic cables in the skull and
brain and limiting movements in animals. This method would
also affect natural behaviors and prevent the observation of
social interactions.
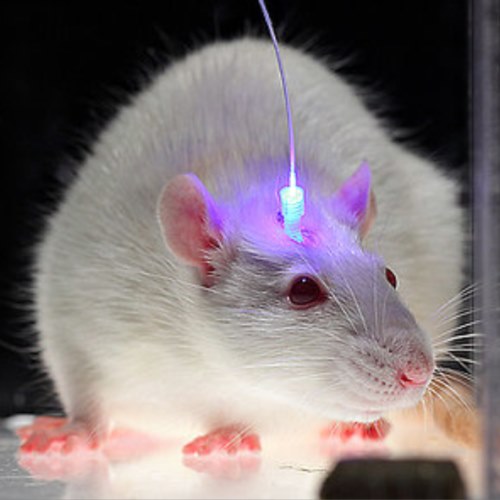
The traditional form of optogenetics used fiber-optic cables and made it difficult to reach deep in the brain.
In a collaborative effort between researchers at the
University of Illinois and Washington University,
advancement in the optogenetics field has been made. An
ultrathin flexible LED, thinner than a human hair, has been
successfully implanted in the brains of mice, allowing
researchers to isolate individual brain functions and
granting them full and precise control over the delivery of
light into the depth of the brain, while the animal is still
fully functional.
“The ideas establish a paradigm for delivering sophisticated
forms of electronics into the body: ultra-miniaturized
devices that are injected into and provide direct
interaction with the depths of the tissue,” said John A.
Rogers, lead researcher and professor of materials science
and engineering at the University of Illinois.
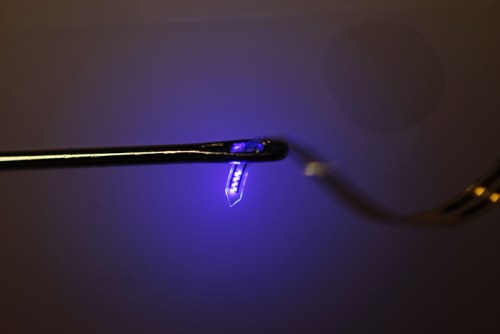
The thin plastic ribbon containing LEDs, electrodes, and sensors printed with advanced electronics is threaded
through the eye of an ordinary
sewing needle.(Image via John A. Rogers)
How it works
The LEDs are printed onto the tip of a thin, flexible
plastic ribbon narrower than a needle head. The devices
include sensors and electrodes that are delivered into the
brain with a thin, releasable micro-injection needle. The
ribbon connects the devices to a wireless antenna and a
rectifier circuit that harvests radio frequency energy in
order to power the devices. Then this module gets mounted to
the top of the head and can be unplugged from the ribbon
when it’s not being used.
Experimenting with LEDs in the brain
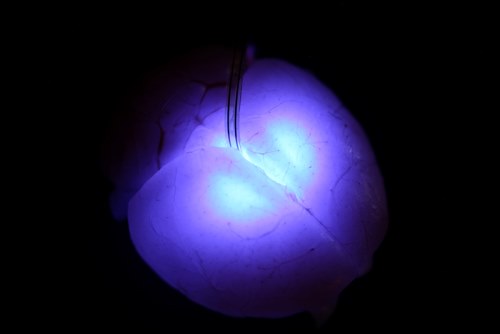
Ultra-miniaturized LEDs injected deep into the brain trigger targeted neurons and provide insight into structure,
function, and complex connections within the brain. (Image via John A. Rogers)
One of the benefits of the new device is that it can be implanted with minimal stress to surrounding tissues. Rogers and his co-researcher Michael R. Bruchas, a professor of anesthesiology at Washington University, experimented with the injectable LEDs in mice and found that they could control certain behaviors by turning individual lights on and off. The experiments revealed that they could train complex behaviors without a physical reward, and could even reduce anxiety responses.
Future impact
In their findings, the researchers have realized that these wirelessly controlled, implantable LEDs can have implications in the treatment of Alzheimer’s, Parkinson’s, depression, anxiety, epilepsy, and other neurological disorders. In addition, technology has the potential to not only be applied to the brain, but other organs as well.
“Many cases, ranging from fundamental studies to clinical
interventions, demand access directly into the depth of the
brain. This is just the first of many examples of injectable
semiconductor microdevices that will follow,” said Rogers.
So who knows what kind of brain manipulation will reveal
itself next, but this may just be the beginning of
injectable brain control and discovering the mysteries
within.