THE METHANE CATASTROPHE THAT AWAITS US
TOMORROW
What happened at the end of the Permian is long, long ago, but not far, far
away. The catastrophic release of methane from the seafloor is not just
something which was long ago. It can, and -- unless we change our way of dealing
with our planet, and change it fast -- will happen again tomorrow.
For most people, global warming -- the gradual and
almost imperceptible increase in the world's average temperature -- is something
that is only a dim and distant threat far in the future...if even then. The
amount of global warming that most of us are likely to experience in our
lifetimes is on the order of a degree or so. Celsius or Fahrenheit (it happens
to be Celsius, in which scale each degree is 1.8 times that of a Fahrenheit
degree): it hardly matters. Such small temperatures changes make no difference
in the lives of most people. After all, the variability of temperature over the
course of a day -- which from where I write is about 17°C (30°F) -- usually
means nothing to us, and whether this spring happens to be a bit warmer or
cooler than last year is typically quite unnoticeable. Moreover, in the affluent
parts of the globe, it is easy to compensate for such minor temperature
differences: with more or less clothing, with a bit more heat during a cold
winter or a bit more air conditioning during a hot summer. Just change the
setting on the thermostat.
The fact that sea level is rising, and that some areas
of the world will eventually be flooded, is known to the better-read members of
the population, but even for them, warming-induced sea level changes seem
remote. Yes, parts of New York City will be flooded in a few centuries, along
with parts of many other coastal cities, but none living today will be around to
witness such flooding. Parts of Bangladesh will go under water, and some island
nations may disappear beneath the waves, but, again, these events will occur in
a distant future to other people in far away lands.
In addition to the indifference caused by our ability
to control our immediate environments, many people have been apathetic about
global warming because they have been told that there is considerable
disagreement among scientists regarding the amount of that warming and exactly
how much is caused by -- and therefore can be controlled by -- human activities.
Unfortunately, these people have been misled, and, in most cases, deliberately
misled. There are always disagreements among scientists, especially those who
are conscientious in their endeavors. That is simply the way that science works.
Disagreement helps determine truth, because it is disagreement that drives
scientists to look for the evidence that will decide the issue. Nature -- the
"real world" -- is always the decision maker.
Regarding global warming -- based on the evidence --
the vast majority of climate scientists are in agreement about three things:
(1) global warming is real, and will continue to increase,
(2) most global warming is due to human activity, specifically the burning of
carbon fuels, particularly fossil fuels (carbon fuels include wood, peat, and
charcoal in addition to the fossil fuels: oil, natural gas, and coal), and
(3) as global warming continues, it will have increasingly adverse effects on
human beings, our environment, and the global economy.
It does not seem worthwhile to review all the enormous
amount of evidence for global warming here. There are many fine books and a
constant stream of scientific papers on the subject. Some of these papers
occasionally attract enough attention that their findings make the daily
newspapers. A few points, however, should be made.
First, the relation between atmospheric levels of the
greenhouse gas carbon dioxide and global temperatures is clear. During the
Phanerozoic (the last 543 million years), when carbon dioxide levels have been
high, global temperatures have been high; when low, global temperatures have
been low. The only exception to this correlation was in the Late Ordovician
(about 440 million years ago), when there was an ice age despite apparently high
levels of carbon dioxide. Other suspected exceptions seem to have been based on
the erroneous presumption that certain fossils that help provide ancient
temperature information were not altered by conditions during and after their
burial, when, in fact, they were. The Ordovician exception has attracted further
investigation. But in the 440 million years since that time, there are no other
exceptions (for a review of their own evidence and that of others, see Royer,
2004). High levels of atmospheric carbon dioxide increase global temperatures
(see Berner and Kothavala, 2001, diagram in previous section).
Second, the current warming is not, as has been
alleged, just part of the natural variation. The current warming, rather, is
quite exceptional. And third, it is due to human activity. Scientists, who
generally are by training (if not by temperament as well) extremely cautious
about reaching conclusions, especially in controversial matters, used to be very
careful about attributing global warming to human activities. With mounting
evidence, that caution has dissipated. In the past few years, the tone of papers
about climate change has shifted. Now scientists refer to anthropogenic
(human-caused) global warming as a matter of course, and many seem quite (and
properly) concerned that their repeated warnings and calls for remedial action
are not being taken seriously enough.
A single graph provides a compelling answer to those
who still disbelieve the connection between human activity atmospheric carbon
dioxide and global temperatures. It traces the amount of carbon dioxide in the
atmosphere over a period of 350,000 years, based on samples of ancient air
bubbles caught in the ice at the Vostok Station in Antarctica, the most
physically remote place on the surface of the planet. There, as snow slowly
accumulated over the ages (Antarctica is technically a desert, because it
receives very little precipitation), the snow also trapped tiny bubbles of air.
From ice cores obtained by drilling through the Antarctic ice, scientists
carefully extract that air, and measure the percentage of carbon dioxide, and
the isotopes of oxygen. The oxygen isotopes can help reveal the temperatures of
long ago. Thus, from the same tiny bubble, scientists can know both the amount
of carbon dioxide in the air, as well as the temperature.
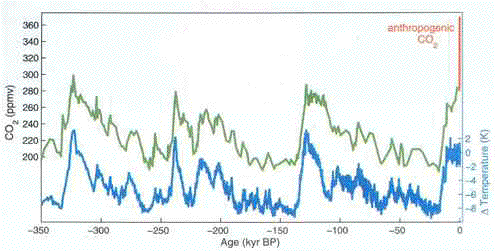
|
| Atmospheric carbon dioxide levels
and global temperatures over the past 350,000 years.
(kyr BP means thousands of years before present.) The scale at the left
refers to the green line, and indicates the level of carbon dioxide in the
atmosphere. Note that it varies from about 200 to 300 parts per million by
volume (ppmv). Temperatures are indicated by
the blue scale at the right, which refers to the blue line. Temperature
variation over the past 350,000 years is about 11°C. (Temperature here is
measured by the Kelvin scale, in which each degree is exactly the equivalent
of a degree Celsius, or C. Each degree Celsius equals 1.8 degrees
Fahrenheit.)
Notice how temperature closely varies with carbon
dioxide level -- except at the extreme right. Here, for emphasis, the green
line for carbon dioxide has been changed to red, to indicate the dramatic
increase in atmospheric carbon dioxide -- in just two centuries. That red
line, incidentally, has reached 380 ppmv as of mid-2005. (Rahmstorf, 2004)
|
The graph shows atmospheric carbon dioxide (CO¸2) and
global temperature rise and fall in lock-step for these hundreds of thousands of
years, right up until the present (the last hundred and fifty years or so).
Then, a vertical red line, marked "anthropogenic CO¸2," abruptly -- and to those
concerned about global warming, alarmingly -- spikes upward. The blue line
designating temperature as yet records no change in response to this surge in
atmospheric carbon dioxide (Rahmstorf, 2004, figure 1), but it will.
Though the red "anthropogenic CO¸2" line spikes
straight up, this reflects the fact that the graph covers a period of more than
a third of a million years; hence, the graph compresses all detail into a
vertical direction. This is useful for comparing the rise and fall of carbon
dioxide and global temperature over long expanses of time, and in putting the
current spike in carbon dioxide in its proper perspective, but it does not
provide insight into the annual changes in carbon dioxide. A different graph,
showing the increase in atmospheric carbon dioxide over the past several
decades, furnishes those details.
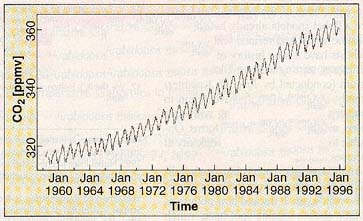
Recent increases in atmospheric carbon dioxide.
(Crutzen and Ramanathan, 2000)
Four things should be noted about this graph. First, it
only records the changes in atmospheric carbon dioxide over the past few
decades. Anthropogenic global warming has been happening since at least the
beginning of the industrial age, about 200 years ago, but scientists only have
precise information from about the past 40 years. Thus, while the graph begins
at less than 320 ppmv (parts per million by volume), the actual pre-industrial
average of atmospheric carbon dioxide is 220 ppmv, as averaged over 420,000
years (Falkowski, 2000). (The immediate pre-industrial level of carbon
dioxide, from about 200 years ago, may have been about 280 ppmv. This value is
commonly employed in climate assessments. Over the past 1000 years, this value
has remained almost constant, with a variation of no more than 10 ppmv.) This
means that the graph omits a major part of the anthropogenic carbon dioxide
increase over the past 200 years, but simply because we were not engaged in the
precision monitoring of carbon dioxide prior to about 40 years ago. As noted
previously, atmospheric carbon dioxide has now reached 380 ppmv.
Measuring Carbon
Dioxide
The amount of carbon dioxide in the atmosphere can be stated in a number of
different ways. The most common way is by indicating the quantity of CO¸2 in
ppmv. The current (as of June, 2005) amount of CO¸2 is over 380 ppmv,
meaning that if we took the entire atmosphere and divided it into a million
equal parts, CO¸2 would constitute 380 of those parts. This quantity could
also have been expressed as a percentage: 0.00038%, but that is such a tiny
percentage that it seems insignificant. Working with such tiny percentages
also tends to cause errors, as when decimal points are misplaced, so the
ppmv value is used instead.
Sometimes scientists employ weight in place of volume. In such cases, the
designation ppmw is used, indicating parts per million by weight. Weight and
volume measures are not identical (think of a cup of air versus a cup of
lead), so scientists need to specify which measuring system they are using.
Another system is that employed for
carbon isotopes, as mentioned previously. This system uses per mil, meaning
parts per thousand. By using per mil rather than percent (%, that is, parts
per hundred), a typical negative carbon isotope shift becomes 3 per mil,
rather than 0.3%. The per mil value is less likely to cause errors, and is
easier to understand and work with. |
Second, the measurements for the graph were taken at
the Mauna Loa Observatory on Hawaii (by Charles Keeling, who figured out how to
measure atmospheric carbon dioxide and is one of the discoverers of global
warming). The Observatory is at the summit of the volcanic peak, which rises
almost 3500 meters (more than two miles) above sea level. Mauna Loa is an
excellent place from which to monitor atmospheric concentrations of carbon
dioxide, as it is far from the most significant sources of that gas; indeed, it
is probably one of the best places in the northern hemisphere from which to
conduct such measurements. (In fact, increased atmospheric carbon dioxide and
global warming were first detected there.) Third, the sawtooth pattern simply
reflects small seasonal variations in the general trend, and so are unimportant.
Fourth -- and quite important -- is that the general trend of the line curves
slightly upward with time (look at the line from its lower left corner). This
means that the problem is getting worse from year to year, not getting better or
merely staying the same.
An additional graph looks at the relation between
atmospheric carbon dioxide and temperature for all samples from which we have
data on both. (The data used in this graph also comes from the ice cores of the
Vostok station.)
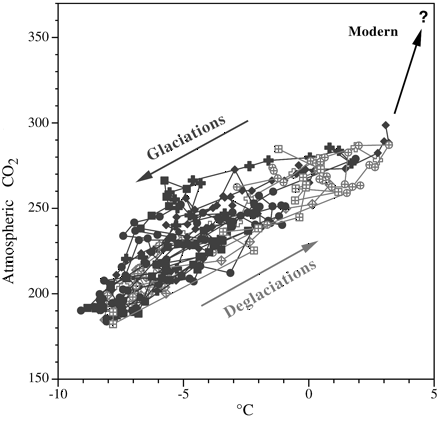
|
| Relation between carbon dioxide
and global warming or cooling over the past 350,000 years.
Each data point (from the Vostok, Antarctica, ice cores) shows both a carbon
dioxide measurement and one for temperature (using oxygen isotope levels).
The data points have been divided into two groups, depending on whether the
data was taken from a time when glaciation was increasing (black points) or
decreasing (gray points). During cooling periods (glaciations), both
temperature and carbon dioxide drop; during warming periods (deglaciations),
both temperature and carbon dioxide rise. The arrows indicate the general
trends. (Falkowski, 2002) |
Each point on this graph displays the information
provided from one air sample, and each is color-coded according to whether it
came from a period of glaciation (black) or deglaciation (gray) during the most
recent part of the Ice Age. Unlike the previous graph, which traces carbon
dioxide and temperature over time, this graph plots the level of carbon dioxide
against temperature, displaying how they have varied in relation to one another.
The arrows show what has happened: the Glaciations arrow indicates that the fall
of atmospheric carbon dioxide correlates with the fall of global temperature.
During Deglaciations, it is the opposite: the rise of carbon dioxide is
correlated with the rise of global temperature.
There is a curious and particularly disturbing aspect
to this graph. That is the direction of the arrow labeled "Modern." Although the
arrows labeled Glaciation and Deglaciation point in opposite directions, they
still define the same linear orientation, rather as a highway defines a
particular route, even though it goes in two different directions. (Like the
hands of a clock -- taking the top of the page as the 12 o'clock direction --
they point in about the 2 o'clock and 8 o'clock directions.) But the "Modern"
arrow veers off on its own (in about the 12:30 direction). It does so because
carbon dioxide is rapidly accumulating in the atmosphere, and is now about 380
ppmv (parts per million by volume).
Global temperatures, however, have not caught up. They
will, and the angle at which the "Modern" arrow veers off will most likely, but
gradually, come to define the same trend as do the Glaciations and Deglaciations
arrows. (In other words, the arrow, like the hand of a clock, will slowly rotate
clockwise from its current 12:30 position towards 2 o'clock as global
temperatures increase.) Unfortunately, unless we severely curb emissions from
the burning of fossil fuels, that arrow will reach to over 560 ppmv of carbon
dioxide (double the pre-industrial level) in the atmosphere before the end of
this century.
Extended to 560 ppmv in the direction currently
indicated, the arrow projects global temperatures may reach about 6.5°C higher
than at present. That projection is equivalent to some of the higher estimates
of the amount the planet will warm by the year 2100, so it does not seem
unreasonable, though many climate scientists believe that warming is likely to
be less. However, if the direction of the "Modern" arrow does with time indeed
slowly rotate clockwise, the planet will be warmer, perhaps considerably warmer,
than those estimates now forecast.
Methane Catastrophe
It is against this background of global warming that a methane catastrophe will
take place. A methane catastrophe consists of a sudden and massive release of
continental margin methane within a short period of time. It is abrupt because
it is initiated by a major submarine landslide, which can happen in a matter of
hours, or by the venting of vast quantities of free and dissociated methane over
a period of decades. These events take place in a geological eyeblink.
Additional slumping and/or venting can continue for centuries to millennia.
The amount of methane that can be released is massive.
Based upon a seafloor temperature increase of 5°C , it is estimated that about
2000 billion metric tons (Gt) of methane could be released (Hornbach, 2004).
(That is one-fifth of the estimated 10,000 Gt of methane hydrate in the world's
continental margins.) There is a simple way to put this amount of methane into
perspective: it contains more than 2 1/2 times the amount of carbon as in the
atmosphere. In addition, methane, it is essential to recall, is over twenty
times more powerful a greenhouse gas than carbon dioxide. Though this methane
would quickly be oxidized -- to carbon dioxide -- in the atmosphere, even its
short-term presence would deliver a stunning jolt of heat to the planet. The
derivative carbon dioxide would maintain that heat over the long-term.
A methane catastrophe, therefore, is an abrupt surge of
greenhouse gas that could make mere carbon dioxide warming of the planet pale to
insignificance. It can utterly overwhelm the natural heat regulatory system of
the Earth, which operates in a much more gradual way, and on a much more
protracted time scale. Its quantity is so massive that there is no remedial
action that people will be able to take to mitigate it except in the most
superficial way. Once a methane catastrophe begins, its consequences for the
planet and its inhabitants, human and other, will be appalling, and we will be
able to do nothing except wait it out.
We are on our way toward such a methane catastrophe. It
will happen because we continue to warm the planet by our burning of fossil
fuels. At the end of the Permian, carbon dioxide was initially injected into the
atmosphere by the Traps eruptions. These eruptions were presumably episodic,
spread out over hundreds of thousands of years, interspersed with long periods
of dormancy. Nonetheless, at least one or more of these episodes was sufficient
to help warm the globe with its carbon dioxide emissions, to directly heat the
Arctic continental margins and cause the release of Arctic seafloor and
permafrost methane, and to trigger the Permian's methane catastrophe.
Though not as dramatically, our releases of carbon
dioxide from the burning of fossil fuels probably exceed, on average, those of
the Siberian Traps. While not episodic, nor as sudden, our own releases are
considerable and increasing. Moreover, our carbon dioxide releases will probably
exceed those of the Traps in a vastly shorter period. In no more than three
centuries, virtually all of the accessible fossil fuel carbon reservoir -- some
5000 billion metric tons (Gt) -- will have been transferred to the atmosphere,
in the form of carbon dioxide. This ought to be quite sufficient to warm air and
ocean enough to liberate a vast quantity of methane from its icy seafloor muds.
Much of the fossil fuel carbon dioxide will also enter
the ocean from the atmosphere, but marine carbon dioxide will not contribute to
global warming (because it is largely out of reach of solar radiation). It will,
however, help acidify the ocean. Excess carbon dioxide is now entering the
ocean, from the atmosphere, at the appalling rate of about a million metric tons
per hour, leading to decrease in ocean surface pH, that is, an increase in
acidity, of about 0.1 pH (Cicerone, 2004). A negative 0.1 pH change may not seem
like much, but it represents a 30% increase in acidity. By the end of this
century, pH will have dropped by another 0.2 to 0.4, possibly increasing acidity
by more than 150%. (Negative pH changes represent exponential [geometric]
increases in acidity rather than linear increases.) Organisms with calcium
carbonate or calcium phosphate skeletons will be hard pressed to survive.
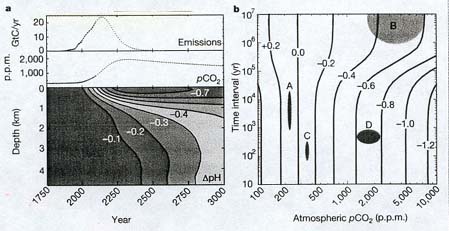
The future acidification of the ocean due to
the influx of carbon dioxide. Diagram a is
composed of three parts. The topmost part indicates the amount of carbon dioxide
which will be emitted by the continued burning of fossil fuels over the next few
hundred years (time scale is on bottom of diagram). Emissions peak about 2150,
and thereafter decline as fossil fuels are used up. Atmospheric carbon dioxide (pCO¸2),
however, increases until it reaches a maximum about year 2250, and then declines
very slowly (the middle part of Diagram a). The bottom part of the diagram shows
the changes in ocean acidity over the next thousand years, according to ocean
depth. Note that it is the surface ocean which is most affected: the negative
figures refer to the change in pH, the measure of acidity. The more negative the
number, the greater the increase in acidity. Diagram b helps put this
coming acidification in perspective, by indicating the change in ocean acidity
over various time scales, from 10 years to 10 million (10^7) years. The blob
labeled A shows the range of ocean acidification during the Ice Age: it was
actually slightly more alkaline (less than 0.0) than today, and its variations
took place over periods from 1000 (10^3) to 10,000 (10^4) years. Blob B shows
the range of ocean acidity over the past 300 million years: acidity varied by up
to 0.6 pH units, but over a very long period of time. Blob C indicates the
minor acid changes over historical time, in the past century or so. Blob D shows
what will happen to ocean acidity over the next centuries, assuming we do not
change our current pattern of fossil fuel consumption: greater acidification
(0.6 to 0.8 pH units) over a vastly shorter period of time than occurred over
the 300 million years indicated by blob B. The basic message of Diagram b: our
continuing burning of fossil fuels will acidify the ocean more -- in just a few
centuries -- than it was acidified during the past third of a billion years.
Human activities will therefore cause the demise of creatures with calcium
carbonate or calcium phosphate skeletons (corals, mollusks, coccoliths, fish,
and lots of other organisms), which will be unable to cope with the rapidly
acidifying conditions. (Caldeira and Wickett, 2003)
A methane catastrophe is just around the corner,
geologically speaking -- as well as in human terms. Assuming we continue to
conduct business as usual, it is inevitable. When it will happen cannot be
predicted, but it will likely begin between about a hundred and, at most, a
thousand years from now. Once it starts -- or even well before it starts -- it
will be irreversible. Each of these characteristics -- inevitability, magnitude,
unpredictability, and irreversibility -- requires further elaboration.
Inevitability
The release of seafloor methane is inevitable because we are pumping
unprecedented quantities of carbon dioxide into the atmosphere. This carbon
dioxide will warm the planet, and, in fact, is already doing so. Though the
amount of global warming thus far (that is, in the twentieth century) is minimal
-- only about 0.6°C (about 1°F), plus or minus 0.2°C -- the warming will
significantly increase during this, the twenty-first century. The most generally
accepted projections for global warming, those from the UN-sponsored
Intergovernmental Panel on Climate Change (IPCC), indicates that the world will
warm by about between 1.4°C and 5.8°C (2.5°F to 10.4°F) by the end of this
century (Kerr, 2001). Though this warming estimate represents the consensus
thinking of the approximately 2500 climate scientists worldwide, recent warming
estimates indicate that it may be too conservative.
Typically, climate scientists make their projections of
global warming by estimating the heating effect of a doubling of atmospheric
carbon dioxide, which has been expected to occur by the end of the twenty-first
century. (The amount of warming that will take place as a result of a doubling
of atmospheric carbon dioxide is often referred to as "climate sensitivity,"
though this is not the precise meaning of the term: see Schlesinger and
Andronova, 2002.) Using a different and complex approach, some scientists now
believe that there could actually be somewhat less warming than projected by the
IPCC.
But these same scientists believe that there is an even
greater likelihood -- in fact, a much greater likelihood -- that warming could
considerably exceed the IPCC's projection. According to their projections,
warming by the end of the century will likely range between 1.0°C and 9.3°C
(1.8°F to 16.7°F), with the upper estimate significantly higher than the IPCC's
estimate. The scientists who reached these conclusions find them -- in a modest
departure from the ordinarily unemotional language of science -- "a disquieting
result" (Andronova and Schlesinger, 2001).
This disquieting result has now (January 2005) been
confirmed as a real possibility by the largest computer climate simulation ever
done. Employing computer time from almost 100,000 home computers, the study
compiled the results from the climateprediction.net experiment.
(The harnessing of huge amounts of home
computer time has become a standard activity in certain branches of science
which require such time for extremely complex calculations. Ordinary citizens
can make important contributions to science by the donation of such unused home
computer time. This endeavor is immensely valuable for climate scientists, and
readers are strongly encouraged to assist. This project does not interfere with
the ordinary use of home computers. Details are furnished at the website,
www.climateprediction.net)
With a doubling of atmospheric carbon dioxide (that is,
the "climate sensitivity"), the study found, the possible global warming could
range from 1.9°C (3.4°F) to as much as 11.5°C (20.7°F). Nonetheless, the study
also found that the most likely temperature increase would be about 3.4°C
(6.1°F), just as the IPCC had (Stainforth, 2005). One of Stainforth's
co-authors, Robert Spicer, pointed out that the highest temperatures in "recent"
earth history occurred some 100 million years ago (during the Cretaceous
Period), but that global temperatures at that time were probably only about 6°C
(10.8°F) higher than today's (Connor, 2005). If the highest likely temperatures
projected by the climateprediction.net study were to come to pass, they would be
without precedent in hundreds of millions of years, perhaps for the entire
Phanerozoic (Royer, 2004).
The disquieting findings of Andronova and Schlesinger
were echoed by Richard Alley of Pennsylvania State University, Chair of the
National Research Council's Committee on Abrupt Climate Change, at the December
2001 meeting of the American Geophysical Union. Alley, who was discussing his
committee's newly released report "Abrupt Climate Changes: Inevitable
Surprises," stated that significant global warming could come much more rapidly
than the IPCC projects. He warned that global temperatures could rise 10°C
(18°F) in just a short time, "tripping the switch" towards abrupt climate change
in only a few decades (Showstack, 2001).
The warming that the planet has already experienced is
not restricted to the lower atmosphere, or to the sea surface. Scientists used
to be puzzled as to where the heat was going, because there seemed to be more
heat being produced by global warming than could be accounted for, based on
atmospheric and sea surface measurements. This is a conundrum no longer. As many
scientists had previously suspected, the heat is going into the oceans. But they
were only able to suspect that the oceans were taking up the heat because they
lacked the ability to measure it. That has changed.
Based on the systematic investigation of millions of
temperature records from various ocean depths worldwide, it is now clear that
the "excess heat" is indeed going into the oceans. In fact, more than 90% of the
heat from global warming has gone into the ocean, with the remaining heat having
gone into the melting of polar region ice and mountain glaciers, and the
atmosphere (Levitus, 2000). The temperature increase (0.06°C, or about 0.1°F) is
minute -- only about a tenth of the temperature rise in the atmosphere -- but it
represents an enormous amount of heat, because of the vast ability of the ocean
to hold heat. The Atlantic, Indian, and Pacific Oceans all record the increase,
and all indicate similar heat variations with time, over a forty year period
from 1955 to 1995. All oceans show a similar increase trend (Levitus, 2000):
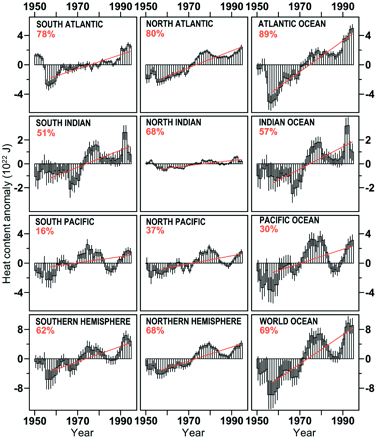
|
| Ocean warming, 1955-1995.
The heat is measured in joules (J), but the specific units are far less
important than the general trends, obvious in all oceans. The red lines and
the red figures indicate the approximate warming that has taken place, based
on millions of measurements. (Levitus, 2000) |
Perhaps most importantly, the warming has penetrated to
deeper parts of all oceans, at depths from 300 to 1000 meters (about 900 feet to
6/10ths of a mile), and in the North Atlantic, even below the 1000 meter level.
The total temperature increase of 0.06°C is the average of temperature readings
down to 3000 meters (Levitus, 2000), emphasizing just how deeply the warming has
penetrated. The North Atlantic data are possibly the most startling, because
that ocean seems to be most vulnerable to global warming, and most able to
impact climatic conditions on its periphery and worldwide, because of its major
role in driving global thermohaline circulation.
As a result of the warming, the ecology of the North
Atlantic seafloor may be changing. The population of small (5 centimeters/2
inches long) marine creatures, the sea cucumbers (holothurians, a large group of
echinoderms and thus the cousins of starfish, sand dollars, and sea urchins) has
increased dramatically. Their numbers have jumped more than a thousand-fold
since just 1996, an increase attributed to an influx of dead algae. Though the
cause of the population surge has yet to be determined, climate change is a
likely suspect (Krieger, 2004).
| The meaning of
catastrophe Catastrophe
is so frequently used in ordinary discourse that it has lost most of its
meaning. Though people in the world's poorer countries often have experience
with catastrophic war, famine, infectious disease and floods (as this is
written, half of Dacca, Bangladesh, a city of 10 million, is currently under
water from monsoon-related flooding, as is 60% of this 140 million
population country), most of the populations of affluent countries have no
experience with catastrophe. What passes for catastrophe are often family
tragedies, which loom large for the affected individuals, but generally
assume no wider significance.
But there are real catastrophes --
sometimes local, sometimes regional, sometimes global -- both for human
beings and the other inhabitants of the planet.
Here is one:
North of Scotland, washed by the
North Sea to the east and the North Atlantic to the west, lie small
archipelagos called the Orkney (about 70 islands) and the Shetland Islands
(about 100 islands). Only a few islands of these, the Northern Isles, are
inhabited, though during the breeding season the islands' rocky cliffs host
huge numbers of seabirds -- guillemots (members of the auk family), Arctic
terns and Shetland kittiwakes (gull family), great skuas and Arctic skuas,
(skua family) -- birds not familiar to most Americans, Europeans, Asians.
These are subarctic birds, which generally live far to the north of the most
populated areas of the Northern Hemisphere (though some of their relatives
do live in more temperate regions).
In recent years, according to
Seabird 2000, the bird count released a few months ago, more than 220,000
pairs of these birds have been breeding in these Scottish islands. But not
this year. In 2004, this huge number of birds produced virtually no young:
at most, a few dozen chicks in all. The breeding season has been a total,
unprecedented failure (McCarthy, 2004). As Subarctic and Arctic regions
warm, the ultimate survival of these birds may be at risk.
This disaster may be the first
ripple of the wave of climate change-induced extinction that will engulf the
planet. The ultimate cause of the breeding failure most probably is the
warming of the eastern North Atlantic, which has pushed warm-water
phytoplankton some 1000 kilometers (600 miles) north -- about a 10° latitude
shift -- in just 40 years (Beaugrand, 2002), and a temperature increase of
about 2°C (3.6°F) in the North Sea over the past 20 (McCarthy, 2004).
(Another source indicates the temperature rise was only 1°C over the past 40
years: Martin Edwards, cited by Proffitt, 2004b.) This warming has resulted
in the northern movement of the plankton that used to live in the
Shetland/Orkney area, as cold-water phytoplankton followed the retreating,
cooler waters. This movement deprived copepods, minute crustaceans which
live off the phytoplankton, of their primary food (Beaugrand, 2002).
The high mortality of the local
phytoplankton and copepods has resulted in a massive die-off of sandeels,
and young sandeels in particular. Sandeels (often written as "sand eels"),
as their name implies, are small (adults from the various species range from
about 20 to 35 centimeter/8 to 14 inch in length), elongated, eel-like fish
that prefer sandy seafloor environments, and burrow into them when
threatened. The sandeels are a major food source for numerous other
organisms.
Although at least one scientist has
proposed that the cause of the plummeting number of sandeels may be an
increase in the local population of herrings, together with the fact that
the North Sea is one of the most overfished ocean areas of the world
(Proffitt, 2004a), this seems rather unlikely. The overfishing has been a
persistent though growing problem; the seabird reproduction failure is
abruptly new. In addition, the population explosion of sea cucumbers,
mentioned previously, confirms that the quantity of organic debris reaching
the seafloor has enormously increased. This debris presumably consists of
the remains of phytoplankton and copepods, whose skyrocketing mortality is
due to oceanic temperature change. Dead phytoplankton and copepods may serve
the dietary needs of sea cucumbers, but the precipitous population decline
of these minute organisms has left the sandeels without sustinance.
Above the sandeels on the food
chain are larger fish, such as cod, whose numbers are falling, marine
mammals, and the birds, now too malnourished to reproduce (McCarthy, 2004).
The former food chain has been replaced by a chain of starvation.
In the far north of Britain, the
great chain of being has been sundered.
[Postscript, 7/12/05: On the
Northwest Coast of the United States (northern California, Oregon,
Washington), there has been a severe decline in the amount of plankton,
including copepods and krill, in the spring and early summer of 2005
(Martin, 2005). Copepods and krill are crustaceans, and (at up to 2.5 cm --
an inch -- in length) are among the largest of the zooplankton, which
consume the vegetative plankton (phytoplankton). Most fish depend on the
copepods and krill for their food supply, directly or indirectly. Local and
migratory seabirds depend on the fish, just as they do in the north of
Scotland. It is not surprising, therefore, that seabirds have borne the
brunt of this new food chain collapse, as they did in the northeast
Atlantic.
In the Farallon Islands, about 40
km (25 miles) off San Francisco, seabird nesting has plummeted. According to
the Point Reyes Bird Observatory director of marine ecology Bill Sydeman,
"We expect zero nesting success" for the Cassin's auklets, a seabird which
breeds on the islands. "We've never seen anything like it" (quoted in
Martin, 2005). Other seabird groups have also been significantly affected, a
situation unprecedented in the thirty years of monitoring. Further to the
north, along the coast of British Columbia and off Alaska, sea surface
temperatures are the highest in fifty years. Along the Oregon coast, these
temperatures are 6°C (11°F) higher than normal (Martin, 2005).
The plankton collapse is attributed
to a major slowdown of upwelling, in which cold water carries nutrients up
from the ocean bottom. Without the influx of nutrients, phytoplankton fail
to thrive and their numbers are greatly reduced. This affects all organisms
above them on the food chain, including fish, seabirds, marine mammals, and
even Humpback and Blue whales (Martin, 2005). Because seabird nesting is
easily observed, its decline is an obvious sign of serious trouble in the
coastal environment. But salmon stocks are down as well -- by a hundredfold
(Martin, 2005).] |
The salinity of the Atlantic Ocean is also changing
(Curry, 2003). Over the four decades between the 1950s and the 1990s, water
closer to the poles has become fresher, and that of the tropics has become
saltier. The more poleward water has freshened because of increasing melting of
the Greenland and Antarctic ice caps and Arctic and Southern Ocean sea ice. The
increase in tropical water salinity is due to increased evaporation: an
additional two meters (yards) of water evaporated over the four decades. This
represents a five to ten percent rise in the evaporation rate in just 40 years
(Curry, 2003).
Both the poleward and tropical changes are
presumptively due to global warming. Indeed, between 50°S (about the latitude of
southern Argentina) and 60°N (the latitude of the southern tip of Greenland),
upper ocean temperatures in the western Atlantic (near the Americas) have risen
about 1.0°C (1.8°F). An increase in equatorial precipitation near the African
coast, possibly also due to global warming, has also been noted (Curry, 2003).
As we have been repeatedly warned by geochemist and
climatologist Wally Broecker (for example, Broecker, 2001), the increasing
freshening of poleward waters, and the increasing salinity of tropical waters,
can slow or shut down the great thermohaline conveyor (for a discussion of
thermohaline circulation, see
APPENDIX 3: THERMOHALINE CIRCULATION). There is, in fact, some evidence of
just such a slowdown, in the subpolar North Atlantic during the 1990s (in
contrast to the late 1970s and 1980s), though its cause is unclear. Lacking
needed data from prior to 1978, when satellite monitoring of the oceans began,
it cannot be determined if the slowing circulation is part of a normal
decade-long cycle, or due to a warming of the water involved in that circulation
(Häkkinen and Rhines, 2004).
A slowdown or shutdown would have enormous consequences
for populations on both sides of the North Atlantic. Though the cooling that
would ensue from a shutdown of thermohaline circulation in the North Atlantic
would be a strictly anomalous regional phenomenon, and could not induce a new
ice age (though some have mistakenly thought so), cooling could nonetheless be
precipitous. In one Ice Age episode, Greenland temperatures fell by 10°C (18°F)
in just a decade (Kerr, 2004). It seems important to re-emphasize that the kind
of North Atlantic cooling projected by Broecker is, paradoxically, the result of
global warming.
As the planet continues to warm, so will the oceans.
The Southern Ocean, at mid-depths, has warmed by 0.17°C (about 0.3°F) since the
1950s (Gille, 2002). New data from the North Pacific confirms that even at great
depths, 5000 meters (three miles) and below, seawater temperature has risen by
0.005°C (0.009°F). This is a tiny change, but it occurs where there should be no
change at all. And it occurs across the entire North Pacific, a distance of many
thousands of kilometers (several thousand miles), from off Washington state to
Japan, as surveyed mostly along latitude 47°N. (The distance is approximately
one-quarter of the circumference of the globe at the latitude surveyed.) And the
warming occurred in just 14 years, between 1985 and 1999 (Fukasawa, 2004).
There should be no change in deep-ocean temperatures
because the deep ocean is quite isolated from the upper ocean, at least on short
time scales. This deep ocean water has not been in contact with the surface in
800 years (Davidson, 2004). This is typical for deep water, although in this
case much of the water ultimately derives from the North Atlantic, and has been
transported to the North Pacific by the great thermohaline conveyor. Textbook
time for ocean mixing -- the amount of time it takes for a mass of ocean water
to blend with other ocean water -- is 1000 years. This slow rate, and the
isolation of deep water from the surface, means that there should have been no
warming whatsoever in the period studied. The fact that there has been such
warming is a matter of surprise and concern -- and even alarm -- to
oceanographers.
The oceans are the roach motel of global warming. In
the case of the oceans, it is heat which checks in but doesn't check out. Water
has an extraordinary heat capacity. In other words, it holds heat better than
almost anything else. That means that when the oceans warm, they lose that heat
only slowly and reluctantly. Ocean heat stays around for a long time. And most
of the heat from global warming -- over 90% -- goes into the oceans.
And the oceans hold 99% of the world's supply of
methane hydrate; the rest is in permafrost. Inevitably, in a warming world,
hydrate methane will also be released from permafrost, but its quantity pales to
insignificance compared to that in the oceans. Moreover, it is not merely
hydrate methane that will be released, but also free methane from below the BSR.
The New BSR
It used to be thought that the depth of the methane hydrates within the ocean
floor sediments would delay and perhaps prevent methane release with global
warming. This thinking was based on the amount of time it would take for warmth
from the overlying ocean to penetrate the sediments, and upon scientific
understanding of the physical contours of the methane hydrate deposits. The
bottom simulating reflectors (BSRs) that marked the boundary between the
overlying hydrate and the underlying free methane closely mimicked the contours
of the ocean floor (hence their name), though generally several hundred meters
deeper.
These contours were fairly smooth and gently rolling
(see the sonar image of the "old" BSR, above, in the Methane and Methane
Hydrates, Part 2, section), and the upper boundary of the hydrate was similarly
presumed to be relatively smooth. Because this hydrate methane was thought to be
located well below the seafloor, it was assumed that heat from global warming
would take a long time to penetrate that far, and that any significant release
of margin methane would take place at least hundreds (Kvenvolden,1988a), and
perhaps thousands of years in the future. Even employing "worst case estimates,"
little methane release was presumed to be possible, because about 98% of methane
hydrate is found in sediment conditions which would require a 4°C (7.2°F)
warming to dissociate (Harvey and Huang, 1995).
Such findings could provide consolation to those
concerned about the possibility of a near-term methane catastrophe, were it not
for the qualifying statement heading a list of conclusions: "In the absence of
fracturing or sediment failure..." (Harvey and Huang, 1995). If fracturing or
sediment failure were indeed possible, the comforting conclusions would not be
valid. In fact, however, fracturing and slumping (sediment failure) turn out to
be the major modes by which large quantities of hydrate methane can be released.
New, more detailed sonar images have completely changed
our understanding of BSR topography. Instead of being fairly smooth and gently
rolling, the BSR surface is now known to be punctuated with sharp, needle-like
peaks, columns, and discontinuous knife-edge ridges, which may extend all the
way up to the top of the sediment. These features apparently represent escape
routes for free and dissociated methane, which at times follows "chimneys" (the
needle-like peaks and columns), and at others follows faults (the knife-edge
ridges) through the consolidated sediments (Wood, 2002; Pecher, 2002). [Sonar
image from Wood, 2002.]
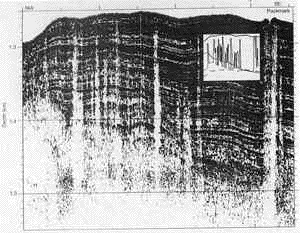
These escape routes are apparently in regular use. If
they were not, we would expect that the free gas below the hydrate would
gradually accumulate and build up pressure. Eventually, the pressure would lead
to a blowout, and explosive release of the gas from its trap under the hydrate.
Though such blowouts may occur occasionally, they are probably not common,
because the faults and chimneys allow methane to escape from below the hydrate.
The escape routes serve as safety valves.
A careful examination of the pressure of the free gas
below the hydrate on several passive margins shows it to be essentially
identical to the pressure needed to pry open overlying faults (Hornbach, 2004).
This pressure is referred to as the critical pressure, and the amount of free
methane under the hydrate is, in passive basins, at critical pressure. Free gas
tends to remain at critical pressure because it forces open the safety valve
when the pressure of the gas exceeds the critical pressure. When the free gas is
at less than the critical pressure, the safety valve remains closed.
Seafloor faults thus seem to be highly responsive as
pressure safety valves. In those active margins where oceanic plates are
subducting beneath continents, the pressure being applied to the wedge of
sediments piled against the continent (the accretionary wedge) forces fluids up
through the hydrates. This process, called hydraulic fracturing, creates
temporary pathways through the sediments, and allows free and dissociating
methane to escape (Zühlsdorff and Spieß, 2004).
Because many active continental margins are being
compressed by the forcing together of tectonic plates, it is not surprising that
faulting and fracturing should be common there. On passive margins, by contrast,
faulting should be much less. Therefore, there should be less free gas, and less
pressure, under hydrate on active margins than on passive ones. And, indeed,
that is the case: on the Blake Ridge off the Florida-Carolina coast, the free
gas column is 200 to 250 meters (yards) thick. Other passive basins display
similar free gas column heights. But the free gas columns in active margins are
much less: typically about 35 meters thick, they rarely exceed 50 meters
(Hornbach, 2004).
Sonar images of an active margin area, off the west
coast of Canada's Vancouver Island, reveal the same sort of wipeout zones
(transparent to sonar: here they are referred to as "blanking zones"; Zühlsdorff
and Spieß, 2004), as reported elsewhere (Wood, 2002). The seafloor surface
exhibits a pockmark, a familiar sign of fluid venting, with massive gas hydrate
lying at only 3 to 8 meters (yards) depth (Riedel, 2002). It is presumably the
process of hydraulic fracturing which allows a rather free flow of methane out
of the hydrate zone, and prevents the buildup to critical pressures found in
passive margins and basins. The process, in fact, may be important for methane
release in all margin settings (Zühlsdorff and Spieß, 2004).
Thus free methane, as well as hydrate, extends in
places to, or almost to, the seafloor, where it can be -- and presumably is --
released into the ocean. This means that at least part of the oceanic methane
reservoir is much more accessible to warming than previously thought. The gas
chimneys and faults that serve as escape routes for methane are therefore likely
to be highly responsive to the oceanic warming that is accompanying global
warming generally. Consequently, assumptions about the relative remoteness and
inaccessability of oceanic hydrates will have to be scrapped. Clearly, methane
hydrates and the free methane below them are considerably more vulnerable to
warming than was previously presumed. Most dismayingly, there may be little lag
time -- or even no lag time whatsoever -- between ocean bottom warming, and the
initiation of seafloor methane release in response.
Magnitude
Initially, methane from hydrate will slowly trickle out of the sediments as the
oceans warm. In fact, some undoubtedly is trickling out now, contributing (along
with other sources such as the increasing number of ruminants and increasing
rice cultivation) to the slow rise of methane detected in the atmosphere. At
some point, however, the gradual mode of methane release is likely to shift to a
pattern of more abrupt, episodic releases, as oceanic warmth penetrates more
deeply into the sediments. There will be rapid depressurization of hydrate at
the base of the hydrate stability zone because of the release of free gas
through warmed chimneys, leading to hydrate dissociation and release. Or there
will be a submarine landslide, triggered by the melting of the hydrate and the
consequent destabilization of deep sediment, or by an earthquake once the
hydrate has been brought to the point of destabilization by the warming.
(A triggering earthquake could be just an ordinary
quake, which are common on active continental margins -- hence their description
as "active" -- or could actually be the result of the warming, which would
increase the weight of the overlying water on continental slopes. Warming
increases the water weight on the continental margins, including the slopes,
because it causes the thermal expansion of water. Thermal expansion means that
the volume of the water increases, but not its total weight, which remains the
same. But since this increased volume is proportionately greater over the
shallower portions of the ocean, the weight of water there increases, while the
weight decreases in the deep ocean where the volume increase is proportionately
smaller. See the diagram of the thermal expansion of water in the Methane and
Methane Hydrates section.)
During the initial phases of more episodic releases,
the rate of methane release will sharply increase. If the release is caused by a
submarine landslide, most of the associated methane will be released in less
than a day. If close to shore, the slide may produce a significant tsunami.
Tsunamis
A tsunami is just another unpleasant possible effect of a submarine landslide.
In 1998, a 7.1 earthquake caused about 4 cubic kilometers (a cubic mile) of
sediment to slide down a 25° seafloor slope a short distance offshore from the
southwestern Pacific island of Papua-New Guinea. A 7 to 10 meter (yard) high
tsunami (perhaps as high as 15 meters/50 feet in the area hardest hit) inundated
the shore just moments later, sweeping away several villages and over 2000
coastal inhabitants (Chang, 2002).

The Great Wave off Kanagawa.
This famous Japanese woodblock print is one of Katsushika Hokusai's "36 views of
Mt. Fuji," and dates from about 1830. That's Mt. Fuji (Fujiyama) in the
background. Kanagawa is a prefecture (district) of Japan, bordering on Toyko
Bay. Although I am not sure whether the wave depicted is actually a tsunami, it
could be: Tokyo Bay is an enclosed bay which could enhance the height of a
tsunami as it crashed toward shore. (From:
www.adachi-hanga.com/hp_english/en_image1/L_003.gif)
Tsunamis caused by submarine landslides are not
uncommon. (The December 2004 tsunami which devastated countries around the
Indian Ocean was not caused by a submarine landslide, but by a magnitude 9.0
earthquake off the Indonesian island of Sumatra. The earthquake caused a huge
movement of the seafloor, and it was this movement which produced the tsunami.)
Usually their effects are confined locally, though this depends on the magnitude
of the slide and its proximity to the coast, among other factors. Larger
landslides do cause bigger tsunamis, other things being equal. And depending on
their size, tsunamis may have regional or even hemispheric rather than merely
local effects.
In November 1929, a 7.2 magnitude earthquake south of
the coast of Newfoundland caused a significant undersea slump which cut a dozen
transatlantic telecommunication cables from North America to Europe. It is
estimated that the slump carried between 300 and 700 cubic kilometers (about 70
to 170 cubic miles) of sediment.
Originating on the continental slope, the Grand Banks
slide tore asunder some half dozen cables, and the turbidity current -- a slurry
of seawater and sediment -- that it engendered ripped apart an additional six.
Every cable was broken in at least two places more than a hundred and sixty-five
kilometers (a hundred miles) apart, indicating both the great width of the slide
and turbidity current, and its speed, estimated at its origin as eighty to one
hundred kilometers (about fifty to sixty miles) an hour. Despite continuously
depositing its load of sand, mud and silt, the turbidity current still retained
enough force to sever a final cable over 800 km (500 miles) seaward (Heezen,
1952).
The height of the resulting tsunami was 7 meters (22
feet). But bays and harbors, because of their constricted shape, tend to channel
tsunamis to more destructive heights as they funnel inland. This fact is
reflected in the name "tsunami," a Japanese word composed of "tsu" meaning
harbor, and "nami" meaning wave. In the case of the Newfoundland tsunami, the
channeling effect caused wave heights to run up to as high as 13 meters (40
feet) in some bays, destroying fishing vessels and harbor buildings and killing
about 28 people (Ruffman, 2001).
Though the Grand Banks slide was probably the result of
the earthquake shaking of waterlogged sediments, some submarine landslides very
likely or certainly have involved free methane gas and methane hydrate, as did
the Storegga slide mentioned previously. The East Coast of the United States
holds its own methane-related hazards, and as scientists have come to know the
marine world better, these hazards have become clearer. The relic of a major
slide, roughly equivalent in volume to that of the 1929 slide off Newfoundland,
has been found off the coast of North Carolina. Estimated to have taken place
about 20,000 years ago, the Albemarle-Currituck slide is of ice age vintage,
though it presumably was not triggered either by the weight of the glaciation or
by glacial rebound, as there was no continental ice sheet within hundreds of
miles.
Recently marine geologists discovered cracks on the
outer edge of the continental shelf (at the "shelf-slope break") along the
Virginia-North Carolina coast to the north of the Albemarle-Currituck slide.
Similar cracks have also been found on the edge of the continental shelf off New
Jersey. On the North Carolina-Virginia shelf edge, these cracks have caused the
shelf edge to slump down as much as 50 meters (160 feet)(Driscoll, 2000). On
closer inspection, however, the cracks have proven not to be simple cracks at
all, but elongated craters as much as two by five kilometers (about 1.2 by 3
miles) in extent (Simpson, 2000).
These craters may be evidence of the rapid, or even
explosive, expulsion of methane-laden fluids from the upper slope sediments, and
could contribute to slope failure, resulting in major submarine landslides. Such
landslides could result in significant tsunamis along the central section of the
East Coast, the discoverers of the Virginia-North Carolina shelf edge cracks
have warned (Driscoll, 2000). A rapid slope failure similar in volume to that of
the Albemarle-Currituck slide could set loose a tsunami up to several meters
(yards) high, equivalent to storm surges from major hurricanes.
As with major storm surges, the actual devastation that
an East Coast tsunami would cause would depend on the topography of the coast
where it hit, together with the height of the tide at the time. (The northern
part of the American East Coast may be particularly vulnerable because it has
numerous estuaries -- Chesapeake Bay, Delaware Bay, New York Harbor -- which can
channel water to major low-lying population centers.) But in important respects
tsunamis differ from the water rise of a storm surge. First and perhaps most
important, there would not be the lengthy warning that accompanies the approach
of a hurricane.
Second, tsunamis are preceded, by several minutes to
about a half hour, by an actual drawdown of sea level of a few to several meters
(yards). This surprising precursor to a tsunami caused several deaths when it
occurred along the French Riviera in response to a submarine earthquake about
thirty years ago. Curious but unsuspecting bathers, lured by the withdrawal of
the Mediterranean and the exposure of sea bottom normally unseen even at the
lowest of tides, wandered out far from shore. There they were caught when the
actual tsunami waves, several meters high, came rolling in a short time later.
This third and last difference between tsunamis and storm surges, that of the
rapid buildup and breaking of walls of water against the coast, rather than the
more gradual rise that accompanies hurricanes, may come several minutes to an
hour after the sea has withdrawn (Driscoll, 2000).
Any continental slope landslide, however triggered, has
the ability to virtually instantaneously release most of the methane from the
landslide material itself. After such a landslide, residual methane from the
landslide scar would be released for weeks to months afterwards. As with
earthquakes and aftershocks, an initial landslide event can trigger additional
slides, generally smaller than the initial slide, but with the non-negligible
possibility of an even greater slide. The amount of the release, obviously,
depends on the quantity of methane that lay below the slide area.
There is no way to predict how large the methane
release from an initial slump might be. This depends on initial conditions on
the seafloor: the stability of the methane hydrate-containing portion of the
continental shelf in one place versus another, the amount of oceanic warming in
specific regions of the ocean, the amount of hydrate and free gas in the
affected area. Oceanic areas close to the poles will be more vulnerable to
methane release for several reasons. First, their stores of hydrate extend to
shallower depths. Second, at shallower depths, sediments are usually of more
recent origin, are less compacted and therefore less stable. Third, global
warming is projected to warm high-latitude (near polar) areas more than mid- or
low-latitude (temperate or tropical) areas, and indeed, it is already doing so.
By the time of an initial methane-related slump,
however, the amount of carbon dioxide in the atmosphere and its resultant global
warming will have well exceeded that needed to trigger the initial event. In
addition, the slump's methane and its successor gas, carbon dioxide, will
contribute to further global warming, and the warming of the oceans will insure
that additional slides will follow. The slides will continue intermittently over
a period of hundreds to thousands of years. Their number and severity will
depend, as with the initial slide, on factors such as slope stability, sediment
consolidation and water content, and regional fault activity, together with the
rate of oceanic warming and changes in global thermohaline circulation and local
currents.
Eventually the slumping will slow and stop, because the
warming of the sediment takes increasing time with increased depth (Nisbet,
1990). As slumping slows, methane input to the atmosphere will also decline,
reducing and eventually removing the source of the warming. By that time, of
course, the use of fossil fuels will have long ago ceased, if not from the
desire to prevent further damage to the planet, then because fossil fuels will
have been completely exhausted. At current rates of use, most of Earth's
petroleum will be gone in fifty years, natural gas in sixty, and even coal, the
supply of which is expected to last some 300 years at current use rates, will be
a distant memory.
Unpredictability
The two possible modes of catastrophically rapid
methane release, slumping and massive dissociation (combined with the release of
the free methane gas which lies below), are each quite unpredictable, as regards
to when each might begin, how long the process might continue, and how much
methane could be released. The problem is compounded by the probability that the
two modes could be combined in many circumstances.
As oceanic temperatures rise, the warmth will first
begin to liberate the free gas and hydrate closest to the sediment surface, in
places (the "escape routes") just a few tens of meters down. Reopening the
escape routes (chimneys and faults), will allow the further escape of free gas
that is buried more deeply under the main bodies of hydrate, hundreds of meters
below. This in turn will depressurize the hydrate at its base, allowing its
dissociation and release. Finally, this increasing dissociation of hydrate will
cause the destabilization of the entire sediment pile, causing slumping.
Just as there is no way to predict how much methane
will be released in an initial seafloor slump, there is no way to determine when
an initial slump will occur. This depends, in part, on how close particular
areas are to threshold conditions. A threshold is literally the sill of a
doorway; it is the point where one enters or leaves a house or room. A threshold
condition therefore is the place where change begins, from one condition to
another. The conditions are often quite different. In the case of continental
shelves, the threshold is between stability and instability, between "just
sitting there" and sliding.
Basically the situation with oceanic slope stability is
no different from snow avalanches, except that we know vastly more about how
snow behaves. Consequently, we employ ski patrols to assess how close mountain
snow is to the threshold where it will let go, and we warn recreational skiers
off slopes when those slopes approach threshold conditions. With oceanic
continental slopes, however, we know vastly less about threshold conditions,
vastly less about the quantity and location of methane hydrate and free methane
that lie within the slopes, and have vastly more area that would require
assessment. Because of the many uncertainties and high cost of such assessments,
it seems highly improbable that they would ever be done.
Irreversibility
The slumping of continental margins due to the warming
of methane hydrate differs in another, extremely important way from snow
avalanches. With snow avalanches, with sufficient warning, not only can we warn
people away, but we can trigger the avalanche ourselves by the use of
explosives. In many mountainous areas there are even artillery emplacements,
from which shells can be fired to cause avalanches to occur when they may do so
most safely. In other areas giant gas burners are used to preemptively trigger
avalanches. These measures can provide some control over avalanches, at least in
those places we monitor and where we take remedial action. With hydrate-related
submarine slides, we have no such option, even if we had the advance warning and
the actual power to trigger such slumps, which we do not. Advance triggering
would only cause the problem we would be trying to avert: the release of methane
from the seafloor. Our one option is prevention: stopping the slumping before it
starts. And that requires that we stop warming the planet.
Once massive dissociation and/or slumping begins, there
will be no way to stop them. Indeed, there will be no way to stop these
processes even from well before they start, because of the lag time between
increasing atmospheric carbon dioxide and the warming of the globe and the
seafloor sediments in which the methane hydrates and free methane reside. As
NASA climate modeler James Hansen has pointed out, "even if rising
concentrations of greenhouse gases could be stabilized tomorrow, gases that have
already accumulated [in the atmosphere] will push surface temperatures up
another half degree or so" (Kerr, 2000), an assessment which is supported by
others (Miehl, 2005).
This amount of additional warming is known as the
"warming commitment" because it represents the amount of warming we are
committed to, even without additional carbon dioxide being dumped into the
atmosphere. Some climate modelers believe the present warming commitment is even
greater than Hansen and Miehl do, and that the planet could warm by another 1°C
(1.8°F) over the next twenty years (Wetherald, 2001) or more (Wigley, 2005). As
time goes on, and carbon dioxide continues to be dumped into the atmosphere, the
amount of the warming commitment will continue to increase.
At some unknown point -- another threshold -- the
amount of carbon dioxide in the atmosphere will be sufficient to eventually
induce the warming of continental slopes enough to trigger enhanced methane
venting, hydrate dissociation, and sediment slumping. Then there will be a
period of time -- more lag time -- before massive dissociation and slumping
actually begin. During this time the atmosphere will be warming, and the oceans
will be warming, and the sediments on the seafloor will be warming. Of course,
that is exactly what is going on now, so it is possible that we have actually
crossed the carbon dioxide threshold needed for these processes to occur, and
that some considerable seafloor methane release is now inevitable.
It is also possible that we have not crossed that
threshold, and may not for many more decades, or even centuries. The time,
however, is short. Commenting on the new determination that climate sensitivity
(with a doubling of atmospheric carbon dioxide) may range from 1.9°C to 11.5°C
(Stainforth, 2005), Oxford University physicist Myles Allen indicated that
"uncertainty over global warming may mean that no such [safe] threshold [for
atmospheric carbon dioxide levels] may exist... 'The danger zone is not
something in the future,' he says, 'We're in it now'" (Hopkin, 2005).
Certainly, based on numerous projections of global
warming, the world will be considerably warmer by the end of this century.
Although the estimates of the size of the temperature increase differ (both the
new Stainforth study and the best IPCC estimate suggest that a 3.4°C [6.1°F]
increase is most likely with a doubling of atmospheric carbon dioxide), all
estimates project an increase. Moreover, it is important to remember that
although most estimates of carbon dioxide release and consequent global warming
project only until the end of the current century (or to a doubling of
atmospheric carbon dioxide from pre-industrial levels, which most models assume
will occur about that time), there is no reason to believe that the
anthropogenic warming of the planet will cease at that point, and, indeed, every
reason to believe it will continue.
The existence of lag time, but of unknown length, means
that we will not suffer the consequences that can ensue when there is immediate
feedback. When someone puts a hand on a hot stove, the message is received
immediately. But when negative consequences do not immediately follow, there is
a tendency to continue behaving the same way as in the past. It is likely,
therefore, that carbon dioxide emissions will continue until there is clear,
dramatic, and unambiguously negative feedback. That is, until catastrophe. Of
course, at that point we will have dumped sufficient amounts of carbon dioxide
into the atmosphere that serious warming will continue for hundreds, thousands,
or even tens of thousands of years, into the future -- even if we then stop the
dumping immediately.
"The added carbon dioxide declines in a markedly
non-exponential manner [that is, not in a smooth, geometric curve]", state the
authors of a section of the 1990 IPCC report (Shine, 1990). "There is an initial
fast decline over the first 10 years period, followed by a more gradual decline
over the next 100 years and a rather slow decline over the thousand year
time-scale. The time period for the first half-life [during which half the added
carbon dioxide will be gone] is typically around 50 years, for the second
[during which half of the remaining added carbon dioxide will be gone], about
250 years" (Shine, 1990).
Calculations such as this have led many to believe that the carbon dioxide we
add to the atmosphere will be mostly gone in just a few centuries, or in a
thousand years at the most. Even the US Environmental Protection Agency (EPA)
states that the lifetime of carbon dioxide is up to 200 years. According to
geophysicist David Archer, however, such projections are in error. His own
calculations indicate that "about 7% of carbon [in carbon dioxide] released
today will still be in the atmosphere in 100,000 years." He further states, "A
better shorthand for public discussion might be that CO¸2
sticks around for hundreds of years, plus 25% that sticks around forever"
(Archer, 2005). Not quite forever, perhaps, but long enough that it could remain
a problem far, far into the future. The carbon dioxide we have already dumped
into the atmosphere, and that which we will dump, in other words, will not
easily go away. Neither will the warming it produces.
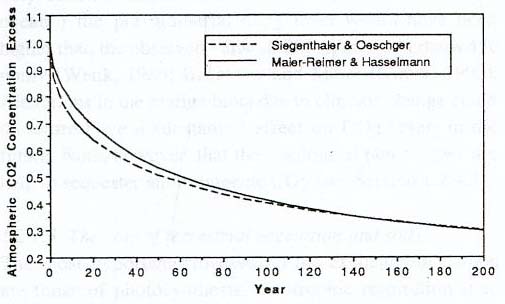
The decline of atmospheric carbon dioxide over
time, based on a doubling of the amount of
carbon dioxide from pre-industrial times. After about 200 years, about 30% of
the added carbon dioxide is still around. Most of the added carbon dioxide winds
up in the ocean, and eventually the additional amount in the atmosphere levels
off to about 15%. [Even after 100,000 years, as much as 7% of the carbon dioxide
now being dumped into the atmosphere will still be around. Because residual
carbon dioxide may stick around for hundreds of thousands of years, and extend
the above curve to the right for several city blocks, it is referred to as the
"long tail" problem: see Archer, 2005.] But because carbon dioxide will likely
continue to be dumped into the atmosphere well beyond the doubling point, this
graph may only provide a rough and possibly quite inaccurate glimpse of what is
to come. (Watson, 1990, based on data from Siegenthaler and Oeschger, 1987, and
Maier-Reimer and Hasselmann, 1987)
Undoubtedly the warming that the planet has experienced
in the twentieth century has already caused the release of additional methane
from permafrost and the continental margins. There is no way that it could be
otherwise: warming releases methane. Furthermore, the rate of methane release
will continue to increase as we continue to warm the Earth. Just when this
release will shift from a more gradual to a more catastrophic mode (from chronic
to acute, to use the medical terms), is unpredictable. This certainly depends on
just how close to threshold conditions continental margin methane is, which is
something we have no way of knowing. (Permafrost methane, although it
potentially could make a major contribution to global warming, is likely to only
be released gradually, though the rate of increase could change rapidly.)
When? How long?
A methane catastrophe can be divided in two parts, the
initial or onset stage, together with that stage's immediate consequences, and
the longer-term consequences.
Onset
For a methane catastrophe to occur, methane must be released in a short period
of time. As mentioned previously (at the start of the Methane Catastrophe
section), scenarios which allow for methane release over long periods (as many
tens of thousands to millions of years) cannot produce catastrophic consequences
because the excess methane would be consumed by expanded populations of
methanotrophs, other essential nutrients being present. To produce catastrophic
consequences, the duration of the initial methane release must be a thousand
years or less, precisely to avoid this sort of methanotroph population
explosion.
Microbes, it should be remembered, can reproduce at
quite extraordinary rates when nutrients are present and conditions favorable.
Under optimal conditions in the laboratory, for example, the gut bacterium E.
coli can double in number about every twenty minutes. Bacterial methanotrophs
presumably have similarly high reproductive rates. While some methanotrophs are
not bacteria, and are -- being archaea -- extremely difficult to cultivate in
laboratories, their potential reproductive rates are also probably quite high. A
gradual, extended increase in ambient methane, therefore, would presumably be
easily consumed by the methanotrophs.
There is a second consideration which suggests that the
onset stage of methane catastrophes cannot be protracted in length. The methane
itself is oxidized in the atmosphere in less than ten years, though large
quantities may temporarily overwhelm the oxidation system and allow for more
extended atmospheric lifetimes. Nonetheless, even with a sudden and massive
release the initial methane would at most be around for only a few decades. Its
successor, carbon dioxide, would remain in the atmosphere considerably longer,
but even the level of carbon dioxide would decline as it is taken up by
photosynthetic organisms and the ocean.
A methane catastrophe wreaks its havoc via three
primary killing mechanisms -- oceanic anoxia, acid rain, and global warming. For
these mechanisms to be maximally effective, they must operate over a very
limited period of time. (As mentioned previously, rate is critical.) With the
level of excess atmospheric carbon dioxide and acid rain starting to decline
just as soon as they are produced (see Watson, 1990, diagram, above, for carbon
dioxide decline), and the global heat the carbon dioxide engenders following
close -- though more slowly -- upon, the faster methane delivers its wallop, the
more powerful it is.
The work of numerous scientists has set an upper limit
on the possible length of the Paleocene-Eocene methane release by their
determinations of the duration of the carbon isotope excursion. Dickens (1995)
found it took less than 10,000 years; Bralower (1997), about 6000 years; Katz
(1999), less than 5000 years; Norris and Röhl (1999) "a few thousand years or
less." Kennett and Stott's 1991 finding that Southern Ocean temperatures jumped
about 8°C in only 2000 years probably further constrains the length of the
Paleocene-Eocene methane release. Considering that the faster it is, the
stronger the punch, therefore, an upper limit of a few centuries to possibly a
thousand years for the initial methane release is not unreasonable. If seafloor
methane was released (at least in part) at the end of the Paleocene and the end
of the Permian via the intrusion of magmatic sills (which must be emplaced on a
time scale of decades: Svensen, 2004), the release time might have been shorter
still.
It should be emphasized that this is simply the
initial, catastrophe-producing release: the initial jolt. As projected here, the
altered climate and oceanic conditions can last for millions of years, as they
did after the initial catastrophic methane release of the end-Permian. This is
because the initial jolt, with its anoxia, acid rain, and global heat, so
reorganizes the global climate and ocean system that dramatically changed
conditions can persist for great lengths of time before and as recovery occurs.
As modeled by Dickens (1997), for example, the residual heat from a major
methane release might last for two million years or so, though the main warming
would quickly follow the release, and only minor warming would persist longer.
Triggers, Present and Past
The same "the faster it is, the stronger the punch" logic that governed the
Paleocene-Eocene warming applies as well to the methane release triggering
mechanism, increasing atmospheric carbon dioxide. With anthropogenic carbon
dioxide, however, carbon dioxide seems to be entering the atmosphere at a rate
vastly faster than with any normal natural process.
We have already consumed almost half of the world's
supply of petroleum. The estimates of the total amount of recoverable petroleum
differ, and some petroleum experts believe that it will be another decade or two
before we reach that halfway point. Nonetheless, within another fifty years,
most of the rest of the world's petroleum will be gone. That means that we will
no longer have to worry about carbon dioxide emissions from petroleum-derived
gasoline and other fuels. (Petroleum is used for other purposes than fuel, such
as for plastics, paraffin, lubricants, and asphalt, but its primary use is to be
burned for energy.) All the carbon dioxide from those emissions will already
have been dumped into the atmosphere, although much will enter the ocean
thereafter. (Fortunately for those who cannot live without the internal
combustion engine, gasoline can be made from coal, as it was by the Germans
during World War II.)
Not only will most of the world's petroleum be gone in
some fifty years, in sixty to seventy years, most of its ordinary natural gas
(that is, excluding that currently locked in permafrost or in continental
margins) will also be exhausted. Each of these carbon reservoirs (petroleum and
natural gas) is estimated to hold about 500 billion metric tons (each roughly
equivalent to an imperial, or American, ton) of carbon (Kasting, 1998). Burned
for their energy and injected into the atmosphere as carbon dioxide, that's more
than enough carbon than is needed to double the atmosphere's current carbon
reservoir (about 750 billion metric tons).
The Earth's reserves of coal, the most plentiful of the
fossil fuels, will last a bit longer: for 220 years, according to some
estimates, for 300 or more, according to others. Obviously, however, and within
two centuries, most of the planet's coal will also have found its way -- as
carbon dioxide -- into the atmosphere and oceans. And there's an estimated 4000
billion metric tons of carbon (Kasting, 1998) that awaits its release via the
burning of coal. That's more than five times the amount currently in the
atmosphere.
For most climate change modeling purposes, future
global warming is estimated on the basis of a doubling of pre-industrial
atmospheric carbon dioxide. Such a doubling is projected to occur, at current
rates of carbon dioxide emissions, before the end of the current century.
(Measurements from the Mauna Loa Observatory now indicate the doubling period
may be closer to sixty than to one hundred years.) Possibly because of the
uncertainties associated with current projections of global warming, few models
examine what could happen as the dumping of carbon dioxide continues beyond the
point where atmospheric carbon dioxide has doubled, but based on our current use
patterns, such dumping is almost certain to continue.
As paleoclimatologist (one who studies ancient
climates) James Kasting has noted (1998), we have the ability not only to double
the pre-industrial level of atmospheric carbon dioxide once, but to double it
again, and double it yet again. (And, it should be added, we could actually come
close to doubling it a fourth time!) And all in a period of perhaps just over
two centuries, or three at most. In short, in addition to the carbon dioxide we
have already dumped into the atmosphere, we have the ability to dump lots more,
and probably will. Enough to thoroughly and rapidly warm the planet, and trigger
a methane catastrophe.
The past 500 million years have recorded only a small
number of methane catastrophes. Those of the end-Permian, the Paleocene-Eocene,
the Toarcian, and perhaps a few of lesser importance in the early Cretaceous may
comprise the entire list. Probably only these satisfy the prerequisite of a
rapid and significant warming trigger.
For the greatest of these events -- those of the
end-Permian, the Paleocene-Eocene, and the Toarcian -- the warming trigger was
presumably twofold. The most important trigger for each of these events was
probably the direct intrusion of seafloor sediments by volcanic magma during the
eruptions which created large igneous provinces: the Siberian Traps, the North
Atlantic Igneous Province (NAIP), and the Karroo Igneous Province, respectively
(Vermeij and Dorritie, 1996; Dorritie, 2002; Svensen, 2004). In addition to
directly heating the hydrate-bearing sediments, the magma also warmed the marine
sediments and, thereby, the ocean. Where the ocean basin was small and
restricted in its circulation, as with the NAIP and the Karroo Igneous Province,
oceanic warming would have been more effective than if the massive volcanism
took place in a less enclosed setting. But colder regions (as with the Siberian
Traps) would have been more seriously impacted than warmer ones, other things
being equal.
Each of these eruptive sequences had a large underwater
component; only the Siberian Traps eruption may have had a major subaerial
(under the air, that is, terrestrial) component as well. But all of these
eruptive sequences injected large quantities of carbon dioxide into the
atmosphere (the Siberian Traps most), the submarine eruptions acidifying the
ocean locally as the carbon dioxide rose through the water column. This
injection of carbon dioxide into the atmosphere caused a general warming of the
surface of the planet, much as we are doing today. Except that our own
(anthropogenic) carbon dioxide release is probably proceeding considerably
faster than the volcanogenic carbon dioxide injections of the past.
Longer-term Consequences
It is certain that the methane that is now being
released from permafrost and the seafloor will contribute to global warming,
initially as methane, with a greenhouse capacity that vastly exceeds that of
carbon dioxide, and then, upon oxidation, as carbon dioxide itself. With a
greenhouse warming ability more than 20 times carbon dioxide, methane that
reaches the atmosphere (and much that is released is likely to do so, because it
will exceed the current capacity of the marine methanotrophs to consume it) has
the potential of doing far more damage to the planet's climate and biosphere
than all the carbon dioxide that has been and will likely be released into the
atmosphere by the burning of fossil fuels.
Simple arithmetic shows why. The total amount of carbon
locked up in fossil fuels is about 5000 billion metric tons (each slightly more
than an imperial ton). As mentioned, in less than 100 years, almost all the
carbon from petroleum and natural gas will have been converted to carbon
dioxide; by about 300 years from now, all the coal will be gone as well. So in
about 300 years, much of 5000 billion metric tons of carbon will have moved into
the atmosphere as carbon dioxide, and about half may then wind up in the ocean,
where it will no longer help warm the planet, but will continue to acidify the
ocean. (There are other natural "sinks" for carbon dioxide besides the ocean,
but research indicates that the absorptive capacity these sinks may change
greatly over time. Consequently, we should not be depending on such natural
sponges for soaking up the carbon dioxide we are pumping into the atmosphere.)
This carbon dioxide -- alone -- contains an amount of carbon equivalent to
almost ten times the amount of carbon that was in the atmosphere at the
beginning of the industrial age, and roughly seven times that of the present.
By contrast, there may be more than 10,000 billion
metric tons of methane carbon that can be released from the seafloor. Though
this quantity is only twice that of the carbon in fossil fuels, it possesses
more than 40 times the short-term warming potential of carbon dioxide. A 'mere'
250 billion metric tons of methane carbon -- less than 1/40th (2.5%) of the
estimated total seafloor methane carbon reservoir -- has the warming capacity of
all fossil fuel carbon. Looked at another way, a release of just 1% of
seafloor methane (somewhat more than 100 billion metric tons) has a warming
potential several times greater than the amount of anthropogenic carbon dioxide
which scientists project will enter the atmosphere in the next 60 to 100 years.
The geological record provides us with only minimal
guidance as to what might be expected from major methane releases. Effects of
the known methane catastrophes (at least those which rise to the level of
"catastrophe," which involves a several degree warming of atmosphere and ocean,
and probably at least transient ocean anoxia), vary considerably. At the lower
end of the range (represented by the oceanic anoxic events of the Toarcian, 183
million years ago, and Aptian, between 116 and 112 million years ago), there was
limited global warming and transient deep ocean anoxia. More serious oceanic
anoxia and warming seems to have occurred at the end of the Paleocene (the
LPTM). Finally, in the most catastrophic event, at the end of the Permian, there
was a stunning global warming, a euxinic ocean that lasted for millions of
years, and the most massive of all mass extinctions.
No doubt variables such as the geographic location of
particular methane-releasing submarine slumps, the general state of the global
climate at the time, and the configuration of continents and oceans played a
role in determining the severity of these catastrophes, but probably the most
significant factors were the amount of and rate at which methane that entered
the atmosphere. While we have no way of estimating how much methane could enter
the atmosphere as a result of current anthropogenic global warming, as noted
above, even the release of a minute proportion of that which is available could
wreak havoc on our planet.
There are too many factors of unknown size to allow any
prediction of the long-term consequences of the coming methane catastrophe. The
end-Permian will not serve as a model, because the world was warmer then, even
before the catastrophe. Furthermore, during the past twenty million years or so,
the globe has cooled considerably. Many climatologists attribute this to the
movement of Antarctica to its current position directly over the South Pole: if
they are correct, the current cooler period of Earth's history will continue
until Antarctica moves off the pole, an event probably tens of millions of years
in the future.
The relatively greater warmth of the Permian period may
have been a contributing factor to the oceanic anoxia of the Early Triassic. It
is possible that in today's cooler world, thermohaline circulation could be
restored quickly, ventilating the deep ocean, and allowing for rapid recovery.
Even so, a "rapid" recovery could take many millennia, perhaps tens or even
hundreds of millennia. If the jolt to the climate system is great enough,
however, and the ocean becomes largely anoxic, methane presumably would continue
to be produced until thermohaline circulation, and therefore deep water
oxidation, are restored.
When will the input of methane overwhelm the global
climate system?
Again, the unknown factors involved prevent a proper
answer. With present-day carbon dioxide, we know the significant sources and
have a good sense of their coming likely concentrations in the atmosphere. There
is no reason to expect that we will be surprised by a major release of carbon
dioxide from an unanticipated source. As destructive as carbon dioxide is -- and
will be -- for the Earth and its inhabitants, at least we have a fairly good
understanding of it, and can -- if we choose -- have some control over it.
This is not the case with continental margin methane.
Although we have a fair estimate of its global quantity, we have little idea of
the details of its worldwide distribution. We have no idea of how close
continental margins may be to the slumping threshold, which presumably varies
from place to place. We do not know how long it will take for the oceans to warm
as the atmosphere does. In fact, we are enormously surprised that they have
warmed as much as they have (Fukasawa, 2004). We thought we knew how fast it
would take for oceanic warming to reach the region of the methane hydrates
(Harvey and Huang, 1995); we were wrong. Ocean warming will release continental
margin methane much faster than we previously thought (Pecher, 2002; Wood, 2002;
Zühlsdorff and Spieß, 2004).
The major factors involved in continental margin
methane release seem to be the following (assuming no major near-term change in
global thermohaline circulation):
1. How fast the oceans are heating up, particularly to the depth of the deepest
methane hydrates (about two kilometers, or 1.2 miles).
2. How fast that heat takes to penetrate the sediments to the base of the gas
hydrate stability zone (BGHSZ). (The BGHSZ, as previously mentioned, is
identical with the BSR, the bottom-simulating reflector detected by sonar.)
3. How much methane will be released through the venting of free gas and
dissociating hydrate as the oceans warm.
4. How close continental margins are to their slumping thresholds.
While we cannot expect to know, except in retrospect,
how close continental margins are to their slumping thresholds (item 4, above),
or how rapidly free methane and dissociated hydrate methane can be released into
the ocean and atmosphere (item 3, above), we may be able to obtain estimates for
the first two factors. The amount of time it would take for ocean warming to
penetrate the sediments to reach the hydrates (item 2, above) has in fact been
estimated. With the findings about the "roughness" of the BSR, and the closeness
of some hydrate and free gas to the seafloor surface (about 15 meters; Wood,
2002), the sediment penetration time may be as short as 55 years for a 6°C heat
pulse (Pecher, 2002). At the other extreme, it could take thousands of years for
a heat pulse to penetrate to the deepest hydrates.
The warming of the oceans may be a second factor that
can be estimated, though we do not yet possess enough data to do so. We do know
that down to a depth of 3000 meters (about 2 miles), the oceans have warmed
0.06°C (about 0.1°F). This is a minuscule amount of warming, but it has been
determined with great precision based on millions of measurements (Levitus,
2000), so it is reliable. We also know that the globe as a whole warmed 0.6°C
(1.1°F) plus or minus 0.2°C (about 0.4°F) during the 20th century (Levitus,
2000). That's 0.06°C ocean warming for 0.6°C global warming. Because the
atmosphere warms first (it has the greenhouse gas) during global warming, and
then it warms the ocean, ocean warming lags atmospheric warming. We therefore
can state with reasonable assurance that the oceans will warm a minimum of about
0.1°C for every 1°C (that's the same ratio as 0.06°C is to 0.6°C) that the globe
warms, at least in the near future.
If we then take the IPCC's maximum estimate for global
warming for the 21st century (remembering that it may be a conservative
estimate), that is, 5.8°C (10.4°F), we can say that the maximum ocean warming in
the 21st century may be 0.58°C (1.04°F). "May be" should be stressed. It could
be less, just as the IPCC's 21st century warming estimate ranges from 1.4°C
(2.5°F) to 5.8°C (10.4°F). On the other hand, it may well be more. Only when we
have more data, in another decade or so, will we be able to know the rate of
oceanic warming with some reasonable certainty.
This small amount of projected deep ocean warming is
unlikely to dissociate much hydrate. At such a slow rate of warming, it would
indeed be several centuries, if not much longer, before even the free gas and
hydrate closest to the sediment surface began to be released. That is, if it
took a significant heat pulse (of say, 6°C, or 10.8°F) to release continental
margin methane. But it does not. At least some free gas below the hydrates,
remember, may be at threshold conditions, right now (Zühlsdorff and Spieß,
2004). That means that any warming whatsoever -- including the tiny amount of
warming which has already occurred -- may be enough to trigger the release of at
least some methane. Like the teapot on the stove in which the water has started
boiling, any increase in global heat can set the whistle blowing -- or the
methane flowing. How much methane will be released is something we will
discover, but in view of the huge amounts of methane available in the
continental margins, even a little may be sufficient to dramatically alter
climate.
Global warming and methane release:
A summary chart
Why the release may begin to arrive sooner than anticipated.
The release of methane from seafloor
hydrate via the warming of seafloor sediments involves a number of processes. As
atmospheric carbon dioxide increases, the atmosphere and surface of the Earth
warm. This warming also warms the ocean. Eventually the ocean's increased warmth
penetrates the sediments to hydrate depth. Estimates have been made about the
duration of each of these processes. But more recent findings or modeling
suggest that the processes may be proceeding more rapidly than the generally
accepted views project:
|
Process |
Generally accepted view |
Cause for concern |
Atmospheric
carbon dioxide increase |
About 100 years to a doubling of CO¸2 from pre-industrial levels (at the
long-term rate of 1.8 ppmv). |
Recent measurements from the climate observatory on Mauna Loa, Hawaii,
indicate that the rate of increase may have accelerated during the past five
years (Lean, 2004). At the current rate (3.0 ppmv), it will only be about 60
years to a doubling of CO¸2. There is no reason to believe, however, that
CO¸2 accumulation in the atmosphere will stop at this arbitrary limit, and
every reason to believe that it will not. |
Warming of atmosphere
("climate sensitivity": The usual way that climate sensitivity is estimated
is with a projected doubling of atmospheric carbon dioxide. Climate
scientists frequently use the formula
DT¸2X
to describe this sensitivity. The
D
means a change in; the T is for global temperature; the
2X
subscript refers to a doubling of
carbon dioxide. ) |
IPCC
says that with a CO¸2 doubling, global temperatures at the end of the
century will likely be in the range of 1.4 to 5.8 °C (2.5 to 10.4°F)
According to Kerr, 2004 (Three degrees of warming), the general consensus
among climate scientists now is that a doubling of atmospheric CO¸2 will
most likely produce a 3.0°C/5.4°F warming. |
Kerr, 2004, does note, however, that while climate scientists generally
agree on a lower bound of 1.5°C/2.7°F for likely climate warming, and a most
probable warming of 3.0°C/5.4°F, there appears to be little agreement on the
upper bound: "The calculation of sensitivity probabilities goes highly
nonlinear at the high end, producing a small but statistically real chance
of an extreme warming." This uncertainty is greatly compounded by the
realization that it seems highly improbable that the anthropogenic increase
of atmospheric CO¸2 will cease with a mere doubling of CO¸2.
In addition, climate modelers Andronova and Schlesinger (2001) foresee a
warming of between 1.0°C and 9.3°C (1.8°F to 16.7°F) by the end of the
century, and Alley has warned that global temperatures could rise 10°C
(18°F) in just a short time, "tripping the switch" towards abrupt climate
change in only a few decades (Showstack, 2001) The climate sensitivity
projected by Andronova and Schlesinger has now received supprort from the
largest climate modeling simulation ever done, the climateprediction.net
experiment involving almost 1000,000 home computers. With a doubling of
atmospheric carbon dioxide, this study indicates that global warming could
be as much as 1.9°C (3.4°F) to 11.5°C (20.7°F; Stainforth, 2005). |
|
Warming of oceans |
Deeper ocean to take about 1000 years to begin to warm |
Levitus, 2000, found that the North Atlantic's temperature, averaged down to
3 km (about 2 miles), has increased 0.06°C, or about a tenth of a degree F,
in 40 years. Similar warming has occurred in all oceans. Fukasawa, 2004,
found a tiny but measurable temperature increase in deep waters (5 km/3
miles) in the North Pacific over a period of 14 years. At such depths, there
should have been no warming at all.
Finally, a slowing of global thermohaline circulation (such as contemplated
by Broecker, 2001) could allow much faster warming in the North Atlantic and
perhaps elsewhere. |
|
Warming of sediments to hydrate release depth |
Thousands of years, based on the former understanding of the BSR
(Harvey and Huang, 1995, and Berner, 2002) Much faster, based on the new
understanding of the BSR (Wood, 2002). |
Pecher, 2002, suggests that heat from a warmed ocean could penetrate the 15
meters of sediment to reach the topmost methane hydrates in as little as 55
years. With much methane hydrate already at critical pressure, methane could
begin to be released from sediment just as soon as it begins to warm
(Hornbach, 2004). Some of the free methane underlying the hydrate, moreover,
may be at threshold conditions, ready for release just as soon as the
sediment begins to warm (Zühlsdorff and Spieß, 2004). |
A DEPLETED EXISTENCE
During a methane catastrophe (there seems no point in trying to describe an
aftermath that lies thousands of years in the future), human beings will face a
depleted existence. Global warming is currently projected to kill off, or
"commit to extinction," between 15 and 37% of presently-existing species in just
50 years (Thomas, 2004); far more will be driven to extinction by the end of the
century, and in the centuries following. Global warming (and its accompanying
effects of acid rain, an acidic and largely anoxic ocean, and so on) will not be
the only cause of the extinction of species: increasing encroachment by the
rapidly growing human population on the habitats of other organisms, the
unceasing exploitation of limited organic resources (as trees, fish) and the
conversion of the planet solely to suit our own needs will also take their
substantial toll. In addition to the extinction of other species, we will also
face the destruction of our own global economy.
There are at least three outstanding examples of what
happens when people ignore the limitations imposed by climate, or when they
simply become the victims of natural climate change.
The Maya
At one time, the Mayan regions of Central America (predominantly in Guatemala
and Mexico's Yucatan) hosted a population of from three to thirteen million.
This was at the height of the Classical Maya, about 750 CE. The Maya, who had
begun to construct cities about 150 CE, apparently suffered one setback about
250 CE, when they abandoned these cities, perhaps as a consequence of drought.
But thereafter the Maya flourished, until the beginning of the ninth century.
Within about one hundred and fifty years, first in more southern and central
regions and later in the north, Mayan civilization fell into sharp decline, and
its cities were abandoned (Haug, 2003).
The probable cause was a series of droughts, the first
short episode occurring about 760 CE. Then, as the area became generally
somewhat drier, three catastrophic multi-year episodes of drought seem to have
happened, centered about 810, 860, and 910 CE. Interestingly, this timing seems
to coincide with periods of intense cold in Scandinavia, possibly indicating a
global rather than regional change in climate. The timing of the droughts also
seems to coincide with a three-stage pattern of the abandonment of Mayan cities,
though this suggestion apparently is controversial. Nonetheless, the episodes of
drought likely gave rise to the cultural upheavals that characterized the
Terminal Classical Period, and the final Mayan collapse (Hodell, 1995; Gill,
2000; and Hodell, 2001, as confirmed by Haug, 2003).
At its peak, Mayan civilization was presumably able to
address the economic needs of its population. But that population, because of
the very success of its economic and political system, probably had reached the
limit of the ability of the Central American region's to provide for Mayan
needs, particularly for food. The food supply depended on the availability of
water, and an assortment of cenotes (natural pools in limestone), reservoirs,
and water conservation strategies were employed to supplement the highly
seasonal rainfall. When climatic conditions began to decline, however, the
region's ability to support the Mayan population also fell, and during the times
of multi-year drought, Mayans would have starved.
The Mayans had reached the carrying capacity of their
environment; then, when conditions became decidedly less favorable, that
carrying capacity plummeted, and with it went Mayan civilization. The term
'carrying capacity' is customarily applied to rangeland in the case of
livestock, and a given ecological area in the case of wild animals. But its
application seems quite appropriate here (it is used by Haug, 2003) in dealing
with the consequences of adverse climate change on human cultures. Numerous
other civilizations -- among them that of the Moche in Peru, the Anasazi in the
US Southwest, that of Ubar in Oman, and many others at the edges of desert
regions in Africa and Asia -- may have suffered similar fates. Significant heat
or cold, or drought, can abruptly lower the carrying capacity of areas inhabited
and developed by human beings, with consequences similar to those that happen to
other creatures.
Iceland
The history of Iceland provides another glimpse of what a depleted existence
could be like. Iceland was settled by the Viking farmers in about 870 CE, and
within fifty years, most of the island's birch trees had been chopped down, for
fuel, building, and clearing land for farming. The farmers had brought cattle,
sheep, horses, goats, and pigs with them, but the goats tore apart the forests
and the pigs ripped up the land in their search for food. Within about two
hundred years, possibly because they had depleted their own food sources, the
goats and pigs had all but disappeared, but the damage they had done remained.
The island's trees were almost gone, and the soil, which requires centuries to
replenish in the colder parts of the world, was rapidly being eroded away
(Ogilvie and McGovern, 2000).
In the meantime, the global climate had turned colder,
and the warm conditions that had allowed the Vikings to settle even in Greenland
were ending. The Greenland settlements were abandoned, or their inhabitants
simply starved, and Icelanders were pushed to the brink. Despite a lack of wood
for fishing vessels, many Icelanders were forced to fish to supplement the
meager production of their farms, which had been devastated by the loss of farm
animals during increasingly harsh winters. Both people and natural climate
change were therefore responsible for the serious economic hardship and even
starvation that occurred in the fourteenth through the sixteenth centuries
(Ogilvie and McGovern, 2000). Those with extensive land and livestock holdings
were in a better position to survive: "It is not surprising that these rulers
probably chose to ignore the early signs of climate change despite its long-term
threats to Icelandic society as a whole: then as now, politics and ecology seem
to have been closely interconnected" (Ogilvie and McGovern, 2000, p. 390).
Easter Island
Another island may provide an even better indication of what happens when people
ignore the constraints their environment imposes. Unlike Iceland, which is a
large island located about a thousand kilometers (600 miles or so) from
Scandinavia, Easter Island is a tiny patch of land (about 160 square kilometers;
64 square miles) isolated by more than 3000 kilometers of South Pacific ocean
from South America, the nearest continent. No ocean-going vessels intentionally
made their way to Easter Island during most of its existence as an inhabited
island (in contrast to Iceland, which was frequented by ships from Scandinavia),
and indeed, for more than a thousand years, the world never knew or even
suspected that Easter Island existed. It has been described as "the world's most
isolated scrap of habitable land" (Diamond, 1995).
Settled in the fifth century by Polynesian seafarers,
Easter Island, like Iceland, had a fertile volcanic soil. Like Iceland, it
possessed a forest, composed, because of its position near the Tropic of
Capricorn (23 1/2°S), of subtropical trees, bushes, and shrubs. Its isolation
made it a haven and nesting site for numerous species of seabirds, as well as
birds like owls, parrots and herons that made Easter home. Shellfish, fish, and
dolphins were abundant locally. The Polynesians themselves also brought chickens
(in addition, inadvertently, to rats), and grew sweet potatoes, bananas,
sugarcane and the edible taro root (Diamond, 1995).
Ultimately, the inhabitants of Easter Island reached a
level of economic stability and success that they were able to devote part of
their attention to the construction of the massive stone figures for which the
island is famous. Hundreds of these huge, multi-ton statues were erected after
being carved out of the rock and hauled long distances (about 10 kilometers, or
6 miles); many hundreds more were abandoned at various stages of completion. The
period of figure carving and statue erection seems to have occurred between
about 1200 to 1500 CE, just as the island's population was peaking at about
7,000 to possibly as many as 20,000 (Diamond, 1995).

Some of the great stone statues at Easter
Island. These statues were constructed and
erected many hundreds of years ago, and thrown down in civil disturbances in the
following centuries. They have been raised again in the 20th century. As they
had in the past, the statues face the empty sea. Were they placed there to warn
off possibly hostile visitors, or, as an earlier version of a Search for
Extraterrestrial (here, off-island) Intelligence, in the forlorn hope that
passing seafarers would contact the islanders?
But the inhabitants were wreaking environmental
destruction on their home. The trees that had been used for construction, fuel,
canoes, and even rope were disappearing within several centuries of the
Polynesians' arrival. By about 1400, the Easter Island palm, its largest tree
and the source of its canoes, was extinct. Dolphins could no longer be hunted:
they were too far out at sea. By the end of the fifteenth century, the forest
was entirely gone. With the destruction of the native vegetation, the soil was
eroded and crop yields declined. Every single species of resident bird was wiped
out, and more than half the seabird nesting sites with them. The size of the
population fell and social order succumbed to lawlessness and thuggery. Even
cannibalism ensued. By about 1700, the population was declining to a mere
fraction (1/4th to 1/10th) of what it had been at its peak. When the first
Europeans arrived, on Easter of 1722 (giving the island its name), they found an
ignorant and impoverished population eking out a subsistence living on a
treeless desert island (Diamond, 1995).
As Jared Diamond (1995) has put it, "Easter Island is
Earth writ small." Our own usually unobtrusive pace of environmental destruction
and climate alteration makes people unaware of -- or, when aware, all too often
indifferent to, or even contemptuous of -- the potential damage of their
activities. But unlike the Easter Islanders, who would not "have noticed the
felling of the last small palm" (Diamond, 1995), scientists have repeatedly
warned that catastrophe awaits us unless we act now to curtail the reckless
exploitation of our planet.
[Note: Since this section was written in 2004, Jared Diamond's book,
Collapse: How Societies Choose to Fail or Succeed, has been published. The
book has received excellent reviews in both Science and Nature.
Though I have not yet had time to read it myself, I have enough familiarity with
Diamond's work that I feel comfortable in highly recommending his book to the
reader.]
The crust of the Earth has been compared to the skin on
the top of a pudding, and ourselves to microbes that live on the skin's surface.
Just as the microbes go about their business undisturbed by the nature of the
skin and the pudding, so we also live on the Earth, taking it for granted, and
largely unaware of its existence.
Below us, and around us, the Earth is in ceaseless
motion. For the most part this motion occurs with extraordinary slowness, as the
great plates of continents and oceans move about the planet's surface and jostle
each other at the rate that fingernails grow. Only when earthquakes smash our
buildings and other constructs, or volcanoes cover cities with ash (as Mount St.
Helens did with Yakima, Washington, in its 1980 eruption), or floods drown
cities, towns and farms (California, North Dakota, Danubian Europe, Germany) as
in the late 20th century and early 21st, do we pay attention, at least for a
bit. But these events typically do not affect us; usually they happen to others
far away, intruding into our consciousness only by way of the morning newspaper
or the evening television news.
Beneath our feet, however, and around us and overhead,
the Earth goes about its own business, in as much ignorance and disregard for us
as we have for it. But if we continue to abuse it, and in particular, if we
continue to use the atmosphere as a sewer (as Stephen Schneider has put it), the
Earth will suddenly and appallingly rise up against us, and we will pay a
terrible, deadly price.
Dan Dorritie
www.killerinourmidst.com
FAIR USE NOTICE
This site may contain copyrighted material the use of which has not always been
specifically authorized by the copyright owner. This material is being made
available as part of the ongoing effort of the scientific community to advance
understanding of environmental, economic, and scientific issues. I believe this
constitutes a 'fair use' of any such copyrighted material as provided for in
section 107 of the US Copyright Law. In accordance with Title 17 U.S.C. Section
107, the material on this site is distributed without profit to those who have
expressed a prior interest in receiving the included information for scientific,
research and educational purposes.
For more information, please go to:
http://www.law.cornell.edu/uscode/17/107.shtml.
If you wish to use copyrighted material from this site for purposes of your own
that go beyond 'fair use', you must obtain permission from the copyright owner.








